In pancreatic α-cells, GLP-1 can reduce the secretion of glucagon, while in pancreatic β-cells, it can promote the secretion of insulin. For GLP-1 to exert these effects, it must bind to its receptor, GLP-1R (Glucagon-Like Peptide 1 Receptor), making GLP-1R one of the crucial targets for diabetes treatment.
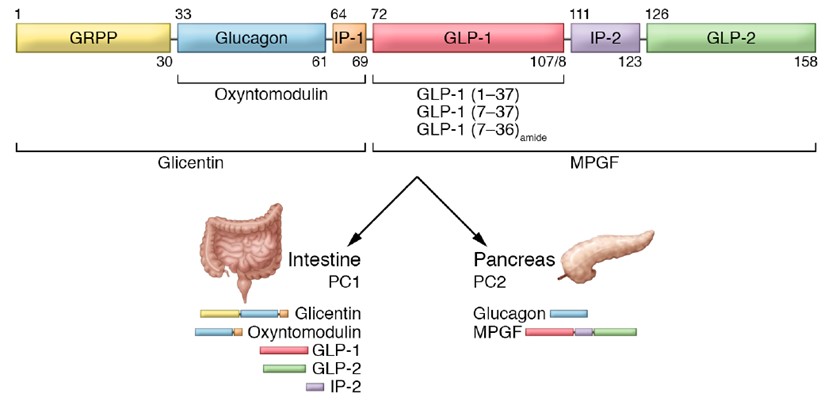
Gene Sequence of Human Proglucagon and Expressed Fragments in Various Organs
(Source: Discovery, characterization, and clinical development of the glucagon-like peptides)
GLP-1R is a class B G protein-coupled receptor (GPCR) with seven transmembrane domains, expressed in the pancreas, lungs, heart, kidneys, hypothalamus, and stomach [4]. As a star target for the development of type 2 diabetes therapeutics, GLP-1R has driven a global upsurge in R&D thanks to the success of its receptor agonists, such as semaglutide and dulaglutide. In China, two independently developed innovative biopharmaceuticals targeting GLP-1R have been approved for marketing and achieved favorable market performance: benaglutide injection by Shanghai Renhui Biotechnology Co., Ltd. and pegylated lixisenatide injection by Jiangsu Hansoh Pharmaceutical Group Co., Ltd.
Pegylated Lixisenatide Injection (Fulaimei) Developed by Jiangsu Hansoh Pharmaceutical Group Co., Ltd.
Benaglutide was approved for marketing in December 2016. It is not only China's first innovative biopharmaceutical in the diabetes field but also the world's first GLP-1 drug with a fully human amino acid sequence. Pegylated lixisenatide, on the other hand, is China's first independently developed long-acting GLP-1 receptor agonist and the world's first pegylated long-acting GLP-1 receptor agonist. The advent of this drug has significantly optimized treatment regimens for diabetes patients: it greatly reduces the frequency of medication administration for patients and largely avoids hypoglycemic events caused by blood glucose reduction. Meanwhile, it also exhibits favorable effects in weight loss, blood pressure reduction, and lipid profile improvement.
A universally recognized challenge in the biopharmaceutical field regarding the R&D of GLP-1R agonists is their short half-life. Although marketed drugs have adopted different formulation strategies to extend the half-life, the overall results show that these drugs still fail to further reduce the frequency of medication for patients. How to achieve a longer half-life and better therapeutic efficacy, or minimize their side effects, remains a difficult problem.
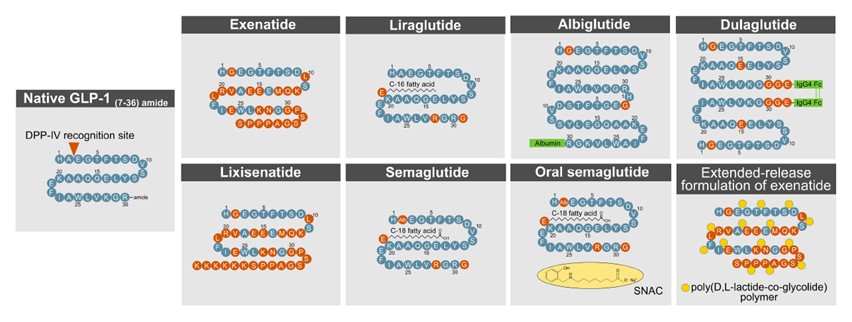
Strategies for Extending the Half-Life of Different Drugs
(Source: Recent Advances in Incretin-Based Pharmacotherapies for the Treatment of Obesity and Diabetes)
Note: Native GLP-1 (7-36) amide contains a DPP-IV (Dipeptidyl Peptidase-IV) recognition site at its N-terminus, which is the main reason for its short in vivo half-life.
Nanobodies are a new type of natural antibody. Their high affinity and targeting ability enable highly specific binding to GLP-1R, achieving more precise drug delivery and therapeutic effects.
Innovative biopharmaceuticals require new drug design concepts. Leveraging the easy modification property of nanobodies, we may design and transform nanobodies into functional fragments that bind to GLP-1R, forming a new strategy. This strategy may repair GLP-1R function to reactivate the receptor, achieve a longer half-life, and even potentially develop drugs that can reduce blood glucose levels while lowering blood lipid or uric acid levels.
A study jointly published by multiple research institutions, including Shanghai Xinpeijing Medical Laboratory and the Department of Laboratory Medicine, Tongji Hospital, Tongji University, Shanghai, designed a new nanobody (sdAb, single-domain antibody) fusion protein named everestmab. In experiments, this nanobody effectively reduced the HOMA-IR (Homeostasis Model Assessment of Insulin Resistance) index in rats, with an efficacy comparable to that of dulaglutide. Additionally, the experiment found that the islet area and pancreatic β-cell mass in rats were improved, which fully demonstrates the effectiveness of nanobodies in drug development targeting GLP-1R [5].
NBLST has been deeply engaged in the nanobody field for more than ten years and has developed a GLP-1R-based nanobody product. Tests show that NBLST's Anti-GLP-1R Nanobody can be used in flow cytometry staining, immunostaining imaging, and ELISA (Enzyme-Linked Immunosorbent Assay). It is inferred that this nanobody can also be applied in immunoprecipitation (IP), co-immunoprecipitation (Co-IP), immunohistochemistry (IHC), and other techniques.
This product can be directly provided to research institutions to effectively detect GLP-1R expression levels, serving as a new research tool for drug development strategies targeting GLP-1R.








